|
These ratios by themselves may not seem particularly interesting or informative however, if we take a ratio of these ratios, we obtain a useful and possibly surprising result: a small, whole-number ratio. (3) Atoms can neither be created nor destroyed. (2) All atoms of the same element are identical different elements have different types of atom. 
It involves the following postulates: (1) Elements consist of indivisible small particles (atoms). 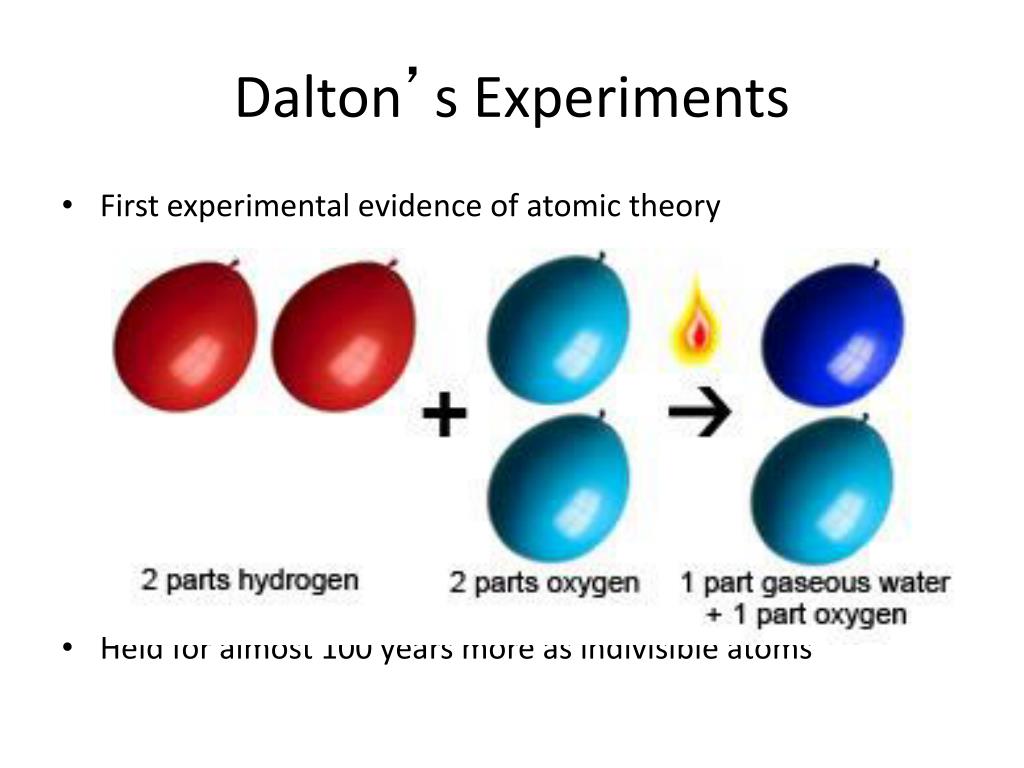
The atoms were unchangeable, indestructible, and always existed. He taught that there were substances called atoms and that these atoms made up all material things. For example, copper and chlorine can form a green, crystalline solid with a mass ratio of 0.558 g chlorine to 1 g copper, as well as a brown crystalline solid with a mass ratio of 1.116 g chlorine to 1 g copper. A theory of chemical combination, first stated by John Dalton in 1803. One of these philosophers was Democritus (460-370 B.C.E.), often referred to as the 'laughing philosopher' because of his emphasis on cheerfulness. The law of multiple proportions states that when two elements react to form more than one compound, a fixed mass of one element will react with masses of the other element in a ratio of small, whole numbers. For example, there are many compounds other than isooctane that also have a carbon-to-hydrogen mass ratio of 5.33:1.00.ĭalton also used data from Proust, as well as results from his own experiments, to formulate another interesting law. According to modern theory, matter is made of tiny particles called atoms, which are in turn made up of subatomic particles. That is, samples that have the same mass ratio are not necessarily the same substance. Atomic theory is a scientific description of the nature of atoms and matter that combines elements of physics, chemistry, and mathematics. If the spheres touch, they are part of a single unit of a compound. The purple spheres represent atoms of another element. 
It is worth noting that although all samples of a particular compound have the same mass ratio, the converse is not true in general. In the following drawing, the green spheres represent atoms of a certain element.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
